Author: Pengcheng(Frank) Shi,Leyu Zhu,Sihan Meng
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Veterinary oral dissolving films (ODFs) are thin polymer matrices designed to disintegrate on the tongue or buccal mucosa of companion and food-producing animals, releasing actives for local or systemic effects without water or forced swallowing. This paper synthesizes the mechanistic pathway—wetting, mucoadhesion, swelling-controlled release, transmucosal transport, and gastrointestinal (GI) uptake—while highlighting species-specific oral physiology, formulation levers, and quality considerations under GMP for veterinary products. We map critical quality attributes (CQAs) to material attributes and critical process parameters (CPPs) and outline an IVIVC strategy suitable for diverse species (canine, feline, equine, small ruminants, and swine).¹²
Keywords: veterinary; oral dissolving films; transmucosal delivery; mucoadhesion; species differences; IVIVC
1. Introduction
ODFs offer practical advantages in veterinary medicine: needle-free dosing, improved caregiver acceptability, and the potential for partial first-pass avoidance via oral mucosal absorption.³ Compared with tablets, ODFs reduce the risk of pilling-related aversion in cats and small dogs and can reduce dose loss due to spitting or regurgitation.² Their thin format and fast disintegration (often ~10–30 s) facilitate dosing in fractious animals and field settings where water is unavailable.¹³
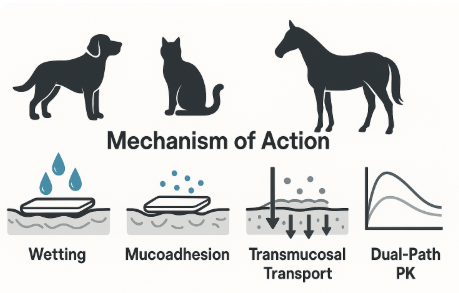
2. Mechanistic Pathway
2.1 Wetting and Film Disintegration
When placed on the tongue or cheek pouch, saliva wets the film, initiating polymer hydration and rapid disintegration. In many species, resting salivary flow varies widely (e.g., dog > cat; equine flow increases with mastication), which alters the local fluid boundary conditions and early dissolution gradients.¹⁴ Formulations therefore target robust wetting across low-saliva states (e.g., anxious cats) and high-flow states (e.g., foals during nursing).
2.2 Mucoadhesion and Residence
Mucoadhesion results from polymer chain interpenetration with the mucus layer, hydrogen bonding, electrostatic interactions, and mechanical interlocking with microfolds.³⁵ Residence time must balance retention (to resist saliva wash-off) with animal comfort and safety; excessive tack risks adherence to gingiva or dental calculus. Carbomers, HPMC, pullulan, alginates, and chitosan derivatives are frequently used to tune adhesion across species.³
2.3 Swelling-Controlled Release
Hydration converts the dry matrix to a gel layer through which actives diffuse toward mucosa. Release rate is governed by polymer grade, plasticizer level, micro-porosity, and residual moisture, which together shape early exposure (useful for rescue or sedation) versus sustained action (useful for local oral care or probiotics).¹³
2.4 Transmucosal Transport
After local release, actives traverse oral epithelium by transcellular diffusion (favored by lipophilicity) or paracellular diffusion (limited for hydrophilic solutes).⁶ Permeation enhancers (fatty acids, bile salts, mild surfactants, chitosan salts) can transiently reduce barrier resistance but must be titrated to avoid irritation or drooling.³⁶ Transporter participation (e.g., nutrient transporters) is molecule- and tissue-specific and can differ by species; thus data from humans do not always extrapolate to cats or ruminants.²⁷
2.5 Dual-Path Pharmacokinetics
A fraction absorbed via the sublingual/buccal route drains to systemic circulation, partially bypassing hepatic first pass; the remainder is swallowed for GI absorption.¹ This often yields a composite PK profile with an early shoulder (mucosal phase) followed by a later GI peak. The balance between the two paths depends on species saliva volume, film placement, residence time, and lipophilicity of the active.²³
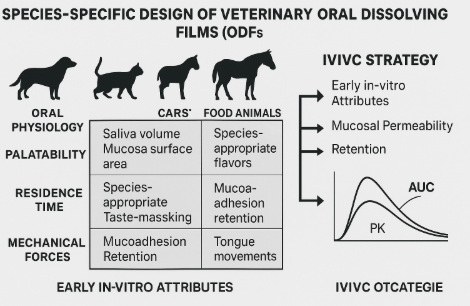
3. Species-Specific Considerations
Canine: Larger oral cavity and typically higher salivary volume support rapid wetting and shorter disintegration; palatability systems (meat flavors, amino acids) improve acceptance but must avoid xylitol toxicity.²⁸
Feline: Lower saliva volume and strong aversion to bitter tastes necessitate intense taste-masking (umami peptides, coated particles) and gentle adhesion to minimize grooming-driven removal.²
Equine: Large mucosal surface and robust salivary flow during chewing favor buccal placement; films must resist mechanical shear from tongue movements; backing layers for unidirectional delivery reduce loss to saliva.⁴
Small ruminants (lamb, kid) and swine: High chewing forces and variable bite behavior demand films with enhanced tear strength and rapid disintegration to limit cud-related loss or spit-out. Field use emphasizes rugged packaging and dosing with gloves.⁴
4. Formulation Levers
4.1 Matrix and Plasticizer
HPMC, PVA, pullulan, alginates, and carbomers provide film integrity and controlled hydration; plasticizers (glycerol, propylene glycol, low-MW PEG) tune flexibility and disintegration.³⁵ For lipophilic actives, solid dispersions, nanoemulsions/SMEDDS, or cyclodextrin inclusion enhance apparent solubility and mucosal partition.³
4.2 Palatability and Sensory
Flavor systems must be species-appropriate (e.g., liver/duck for cats, poultry/beef for dogs, apple/molasses for equine) while preserving disintegration. Sweeteners must avoid species-specific toxicities and dental concerns.²⁸
4.3 Permeation Modulation and pH Microenvironment
Weak acids/bases benefit from micro-pH buffering in the film to maximize the unionized fraction at the mucosal interface. Enhancers are selected to meet oral tolerability thresholds validated in target species.³⁶
4.4 Target CQAs
Disintegration time (~10–30 s), content uniformity per piece, tensile/tear strength, residual solvent/moisture, surface pH, microbial limits (non-sterile oral), palatability acceptability scores, and in-vitro mucosal permeability (P_app) on species-relevant tissues.¹³
5. In-Vitro and In-Vivo Assessment (IVIVC)
In-vitro: Simulated saliva (species-adjusted pH, ionic strength, mucins), dynamic disintegration tests under shear representative of tongue motion, and permeability using ex-vivo canine/feline/equine buccal tissues or validated 3D oral epithelium models.⁶⁷
In-vivo: PK endpoints ((C_{max}), (T_{max}), AUC) versus oral solution/tablet comparators in the target species; user-centric endpoints (ease of dosing, spit-out rate, drooling, time-to-calm for sedatives) recorded by trained handlers.²⁴
IVIVC concept: Early in-vitro release fraction + P_app + residence time are regressed to (T_{max})/AUC to build predictive relationships guiding dose selection and scale-up.¹³
6. Manufacturing and Quality
An ODF production train includes solution preparation, filtration/deaeration, precision coating (slot-die/gravure), staged drying/conditioning, slitting and die-cutting, and high-barrier unit packaging.¹ Quality-by-Design (QbD) links material attributes (polymer grade, plasticizer %, enhancer level) and CPPs (coat weight, oven profile, web tension) to CQAs. PAT (in-line thickness, NIR/Raman for solids/API, vision inspection) supports batch release.³⁸ Veterinary GMP also requires species-specific stability (palatability drift, aroma loss) and label controls for food animals (withdrawal times, residue limits).⁴
7. Safety and Welfare
Films must avoid oral irritation, excessive tack, choking risk, and dental adherence. Enhancers and flavors are screened for mucosal tolerability and species toxicity (e.g., no xylitol for dogs/ferrets).²⁸ For food-producing species, excipients and actives must conform to residue tolerances and withdrawal periods.⁴
8. Use-Case Archetypes
Rapid behavior control or rescue scenarios (e.g., when fast onset is desirable): prioritize fast disintegration, moderate mucoadhesion, and high partitioning toward mucosa.¹²
Chronic daily supplementation (vitamins, oral care actives, probiotics): emphasize palatability, consistent residence, and low sugar/energy burden.³
Large-animal field dosing: rugged films and unit pouches resistant to humidity/temperature swings, with pictorial instructions for quick placement.⁴
9. Conclusions
Veterinary ODFs leverage a coupled sequence—wetting, mucoadhesion, swelling-controlled release, transmucosal transport, and optional GI uptake—to deliver actives rapidly and humanely across species. Success depends on species-aware formulation (matrix, palatability, enhancers), precise R2R manufacturing under QbD/PAT, and IVIVC linking in-vitro attributes to in-vivo exposure. By aligning CQAs with target species behaviors and welfare constraints, ODFs can expand therapeutic options and adherence in both companion and production animals.¹²³⁴⁶⁷
References
¹ Dixit RP, Puthli SP. Oral strip technology and film manufacturing principles. J Control Release.
² American & European animal-health guidance on oral transmucosal dosing and caregiver acceptability (companion animals).
³ Preis M., et al. Oromucosal delivery, mucoadhesion, and patient-centric film design. Eur J Pharm Biopharm.
⁴ VICH/FDA/EMA veterinary GMP and stability expectations, including food-animal residue/withdrawal considerations.
⁵ Smart JD. Fundamentals of mucoadhesion for transmucosal systems. Adv Drug Deliv Rev.
⁶ Nielsen HM, Rassing MR. In-vitro models for buccal permeability and species considerations. Eur J Pharm Sci.
⁷ Comparative oral physiology sources on canine/feline/equine mucosa and salivation (veterinary dentistry/pharmacology texts).
⁸ Web-coating and PAT monographs for pharmaceutical films (slot-die thickness control, NIR/Raman content monitoring).