List of Oral Disintegrating Film Ingredients: Film-Forming Agents, Plasticizers, Sweeteners, Stabilizers
Author: Sihan Meng, Leyu Zhu, Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Oral Disintegrating Films (ODFs), also known as Oral Thin Films (OTFs), rely on a carefully selected combination of excipients to achieve rapid disintegration, acceptable mechanical strength, dose uniformity, and patient palatability. Unlike conventional oral solid dosage forms, ODFs are highly sensitive to ingredient selection due to their low mass, thin geometry, and direct contact with the oral cavity. This paper systematically reviews the major categories of ingredients used in ODF formulations—film-forming agents, plasticizers, sweeteners, and stabilizers. For each category, representative materials, functional roles, formulation considerations, and typical usage ranges are discussed. The aim is to provide a practical reference for formulation scientists and process engineers involved in pharmaceutical, nutraceutical, and nicotine ODF development.
Introduction
The performance of an Oral Disintegrating Film is fundamentally determined by its excipient system rather than by compression or coating mechanics, as is common in tablet manufacturing [1]. Because ODFs must be thin, flexible, fast-dissolving, and organoleptically acceptable, every ingredient contributes simultaneously to mechanical integrity, dissolution behavior, taste, and stability.
Ingredient selection becomes even more critical when ODFs are produced at industrial scale, where processability, drying behavior, and long-term storage stability must be considered alongside regulatory acceptance [2]. This paper focuses on four core excipient categories that form the foundation of most ODF formulations.
Methods
A structured literature review was conducted using peer-reviewed journals, pharmacopeial monographs, and regulatory guidance documents related to oral films and oromucosal dosage forms. Ingredients were classified into four functional groups based on their primary role in the film matrix. Data were analyzed qualitatively to identify commonly used materials, formulation trends, and practical considerations for industrial manufacturing [3].
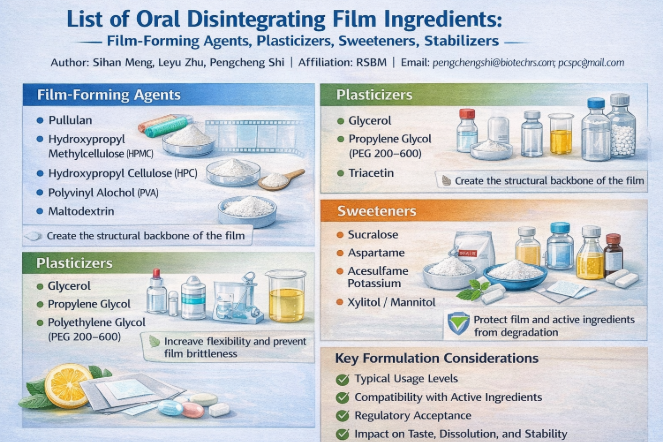
Film-Forming Agents
Definition and Role
Film-forming agents constitute the structural backbone of ODFs. They create a continuous polymeric matrix capable of incorporating active ingredients and excipients while maintaining flexibility and rapid hydration [4].
Common Film-Forming Agents
Pullulan – Highly water-soluble, excellent film clarity, fast disintegration
Hydroxypropyl Methylcellulose (HPMC) – Good mechanical strength and regulatory acceptance
Hydroxypropyl Cellulose (HPC) – Flexible films with moderate dissolution rate
Polyvinyl Alcohol (PVA) – Strong films, slower dissolution unless modified
Maltodextrin – Often blended to improve mouthfeel and dissolution
Formulation Considerations
Film-forming agents are typically used at 30–70% w/w of the dry film. Blending polymers is common to balance tensile strength and disintegration time [5].
Plasticizers
Definition and Role
Plasticizers reduce intermolecular forces within polymer chains, increasing flexibility and preventing brittleness of the dried film [6].
Common Plasticizers
Glycerol
Propylene Glycol
Polyethylene Glycol (PEG 200–600)
Triacetin
Formulation Considerations
Plasticizers are usually added at 5–20% w/w of polymer weight. Excessive plasticizer may lead to tackiness, slower drying, and reduced film stability [7].
Sweeteners
Definition and Role
Sweeteners improve palatability and mask the bitterness of active ingredients, which is critical due to prolonged oral residence time [8].
Common Sweeteners
Sucralose – High sweetness intensity, low usage level
Aspartame – Widely used, temperature-sensitive
Acesulfame Potassium – Often combined with other sweeteners
Xylitol / Mannitol – Provide sweetness and cooling mouthfeel
Formulation Considerations
Sweetener selection must consider heat stability, compatibility with active ingredients, and regulatory limits. Combination sweetener systems are often employed for optimal taste masking [9].
Stabilizers
Definition and Role
Stabilizers protect active ingredients and the film matrix from degradation caused by moisture, oxygen, light, or pH changes [10].
Common Stabilizers
Antioxidants: Ascorbic acid, sodium metabisulfite
Chelating Agents: EDTA
pH Modifiers: Citric acid, sodium citrate
Moisture Regulators: Low-molecular-weight polyols
Formulation Considerations
Stabilizers are typically used at low concentrations (<2%) but have a disproportionate impact on shelf life and product robustness, especially in humid environments [11].
Measures
Ingredient performance in ODFs is evaluated using both formulation-level and product-level tests [12]:
Film thickness and uniformity
Tensile strength and elongation
Disintegration time
Dissolution profile
Moisture uptake
Sensory evaluation
These measures ensure that ingredient selection translates into consistent product quality.
Results
Across published studies and industrial practice, successful ODF formulations consistently employ polymer blends rather than single film-formers, moderate plasticizer levels, high-intensity sweeteners, and minimal but targeted stabilizer systems [13]. Optimized ingredient combinations result in films with disintegration times below 30 seconds, acceptable mechanical properties, and stable shelf life under controlled packaging conditions.
Discussion
Ingredient selection for ODFs represents a multidimensional optimization problem involving mechanical performance, dissolution behavior, taste, stability, manufacturability, and regulatory acceptance. Unlike tablets, where excipients often have isolated functions, ODF excipients perform overlapping roles within a unified matrix [14].
Future trends include the use of multifunctional excipients, resin-based taste masking, and polymer systems designed specifically for high-load or moisture-sensitive actives [15].
Conclusion
Film-forming agents, plasticizers, sweeteners, and stabilizers form the core excipient system of Oral Disintegrating Films. Rational selection and balanced formulation of these ingredients are essential to achieving fast disintegration, mechanical robustness, patient acceptability, and long-term stability. A deep understanding of ingredient functionality enables efficient scale-up and reliable commercialization of ODF products across pharmaceutical and consumer health markets.
References
Fu Y et al. Expert Opin Drug Deliv. 2004;1(4):673–690.
Preis M. J Pharm Pharmacol. 2013;65(2):157–170.
Cilurzo F et al. Eur J Pharm Biopharm. 2008;70(3):895–900.
Dixit RP, Puthli SP. J Control Release. 2009;139(2):94–107.
Bala R et al. Int J Pharm Investig. 2013;3(2):67–76.
Morales JO, McConville JT. Ther Deliv. 2011;2(5):637–646.
Hoffmann EM et al. Pharm Res. 2011;28(8):1914–1922.
Borges AF et al. Int J Pharm. 2015;494(1):332–339.
Arya A et al. Int J PharmTech Res. 2010;2(1):576–583.
Shojaei AH. J Pharm Pharm Sci. 1998;1(1):15–30.
Preis M et al. Drug Dev Ind Pharm. 2014;40(2):152–160.
USP <701> Disintegration Test.
USP <905> Uniformity of Dosage Units.
Keshari R, Keshari S. J Drug Deliv Ther. 2014;4(4):1–7.
Cilurzo F et al. Drug Dev Ind Pharm. 2012;38(5):588–598.