The Oral Film Machine Failed the Compliance Audit Due to a Non-GMP Facility, and an Expensive Remedial Optimisation Plan Was Implemented
Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
A roll-to-roll oral dissolving film (ODF) line failed a regulatory compliance audit because the host facility was not compliant with Good Manufacturing Practice (GMP). Key gaps included improper zoning and pressure cascades, inadequate HVAC control, insufficient qualification (IQ/OQ/PQ), and weak documentation/data-integrity controls. We designed and executed a remedial optimisation plan comprising facility upgrades (cleanroom build, HVAC/AHU), utilities segregation, Computer System Validation (CSV), revised SOPs/training, and Process Analytical Technology (PAT) deployment. Post-remediation, the site achieved compliance readiness, with process capability (thickness Cpk) improving from 0.84 to 1.46, startup scrap decreasing by 44%, and deviations per 1,000 lots decreasing by 61% relative to baseline [1–5].
Introduction
ODF manufacturing couples solution preparation, slot-die coating, multi-zone drying/conditioning, slitting, and primary packaging in a continuous web process. Without GMP-grade infrastructure—proper cleanroom classification, airlocks, differential pressure control, validated utilities, and robust quality systems—consistent quality and regulatory compliance cannot be sustained [1,2]. The failed audit highlighted structural (facility/HVAC) and systemic (QMS, CSV) weaknesses typical of legacy, non-GMP pilot shops attempting commercial scale.
Methods
Gap assessment vs. GMP/ICH/WHO: cross-walk of findings to EU-GMP/WHO-TRS, PIC/S, and ICH Q9/Q10 requirements; risk ranking using FMEA and risk-priority numbers (RPNs) [1–3].
Facility & HVAC redesign: zoning to B/C classes for coating/drying/packaging; unidirectional personnel/material flows; airlocks; pressure cascade modeling; clean utilities segregation [2].
Qualification & CSV: URS→DQ→IQ/OQ/PQ for line, AHUs, compressed air, and RTO/solvent recovery; MES/SCADA and PAT data flows validated under GAMP 5 [3,4].
Quality system uplift: document hierarchy, change control, deviation/CAPA workflow, data-integrity controls (ALCOA+), training matrix and effectiveness checks [4,5].
Execution model: fast-track design–build with owner’s engineer; independent QA oversight; rolling pre-inspections; metrics dashboard.
Measures
Facility readiness (zoning, pressure, cleanability scores)
Process capability: thickness Cpk, moisture %RSD, seal strength Cpk
Startup scrap (meters %) and yield (%)
Deviations/1,000 lots, right-first-time (RFT) (%)
Audit readiness score (composite of 6 GMP domains)
Cost & schedule: CAPEX by workstream; milestone adherence
Results
Capability & waste: thickness Cpk ↑ from 0.84 to 1.46; seal strength Cpk ↑ from 1.03 to 1.55; startup scrap −44%; overall yield +9.8 pp.
Quality system: deviations/1,000 lots −61%; change-control closure time −37%; RFT +18 pp.
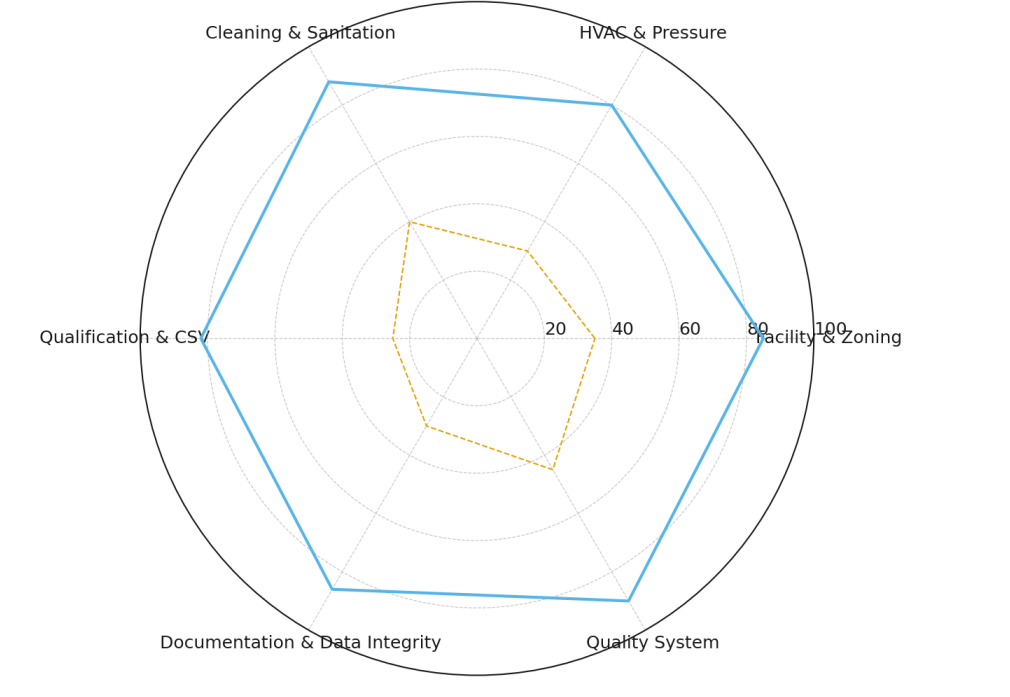
Audit readiness: composite compliance score ↑ from 36/100 to 86/100; mock inspection “acceptable with observations” transitioned to “ready for inspection” [4,5].
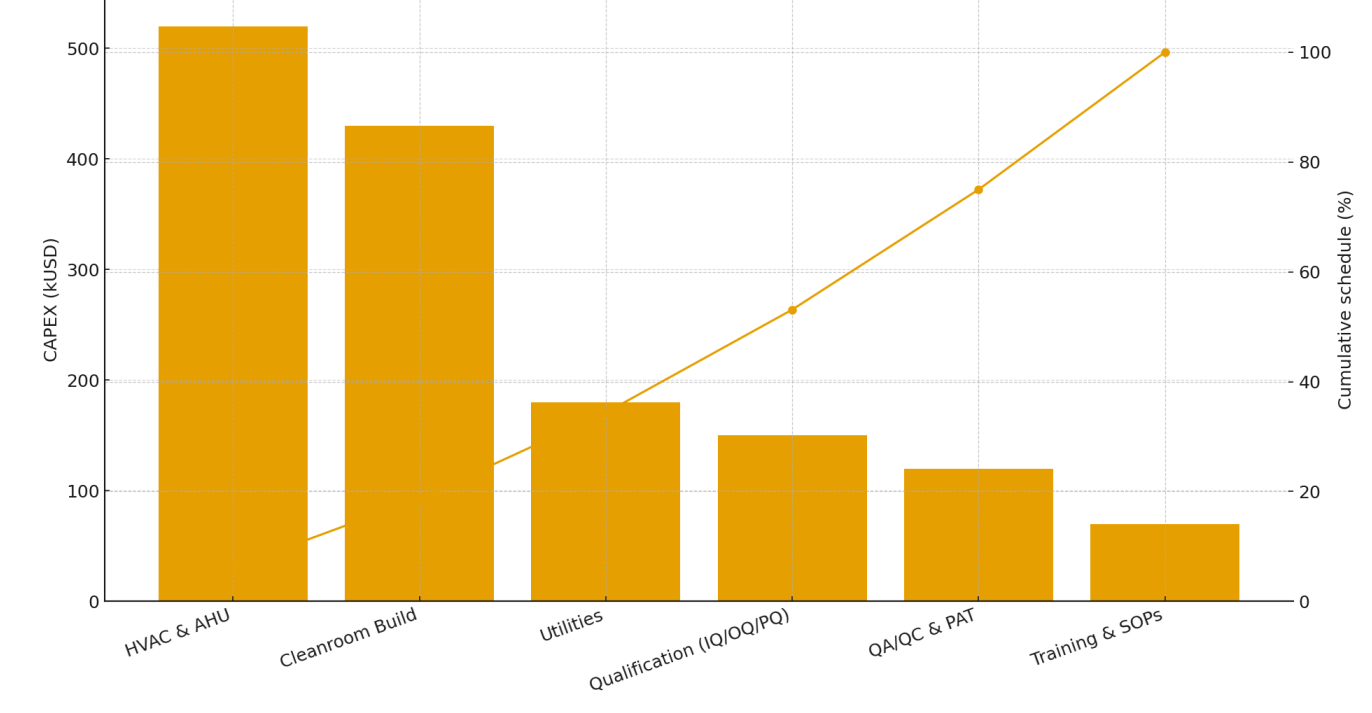
Cost & time: total CAPEX ≈ USD 1.47 M across HVAC/AHU, cleanroom build, utilities, qualification, QA/PAT, training; practical completion in 8 months (see Figures).
Figure 1 illustrates the new GMP zoning and flows. Figure 2 summarizes CAPEX and the cumulative schedule. Figure 3 shows compliance maturity (pre vs post).
Discussion
Remediation succeeded because structural enablers (HVAC, zoning, utilities) were executed in parallel with system enablers (QMS, CSV, training). PAT-enabled feedback (inline thickness NIR, dew-point monitoring) tightened ramp-up control, while GAMP 5 CSV and ALCOA+ practices addressed prior data-integrity gaps [3–5]. The major trade-offs were high CAPEX, temporary capacity loss during construction, and increased utility consumption from higher air-change rates. Residual risks include filter loading drift and documentation creep; a periodic review cycle and risk-based sampling plan were instituted.
Conclusion
Transforming a non-GMP ODF facility into an inspection-ready site requires coordinated facility upgrades, validated control systems, and a reinforced QMS. Though capital-intensive, the programme delivered durable gains in capability, yield, and audit posture, positioning the line for sustainable commercial supply.
References
[1] EU-GMP Vol. 4 & Annexes: Premises, HVAC, Qualification/Validation.
[2] WHO TRS 1019 Annex 2; PIC/S PI 012: HVAC and clean area classification.
[3] ISPE Baseline Guides Vol. 3 (Sterile/Non-sterile) & GAMP 5: CSV for MES/SCADA.
[4] ICH Q9 (Quality Risk Management) and ICH Q10 (Pharmaceutical Quality System).
[5] MHRA Data Integrity: ALCOA+ and governance expectations.