Non-GMP ODF Facilities Failing Compliance Audits: Issues and Optimisation Solutions During Costly Remediation
Author: ZHULEYU (RSBM)
Abstract
Oral dissolving film (ODF) manufacturing lines operating in non-GMP facilities frequently fail regulatory audits due to gaps in quality systems, facility/utility controls, data integrity, and environment-dependent packaging. We describe typical findings, construct a remediation roadmap, and quantify the operational and economic impacts of a structured optimisation program. Using pre/post audit tallies, capability and stability metrics, and cost–benefit modeling, we show that comprehensive remediation—though expensive—reduces critical/major findings >80%, shortens batch release cycle time, and restores contract manufacturing eligibility. Figures visualise audit finding distributions, a remediation Gantt timeline, and a cost–benefit waterfall [1–7].
Figures (landscape, low memory)
Introduction
ODF production is a roll-to-roll process sensitive to utilities (HVAC, pressure cascades), cleanliness classification, data governance, and packaging atmospheres. Non-GMP facilities often rely on ad-hoc SOPs, unqualified equipment, and weak change control, which translate into audit failures and commercial interruptions. We propose a GMP-aligned remediation sequence that stabilises product CQAs (thickness, residual solvent, moisture, seal integrity) and meets inspection expectations from FDA/EU-GMP/PIC/S [1–4].
Methods
Gap assessment: cross-functional audit against EU-GMP Vol. 4, Annex 1/15, and PIC/S PE 009; risk ranking with severity (Critical/Major/Minor).
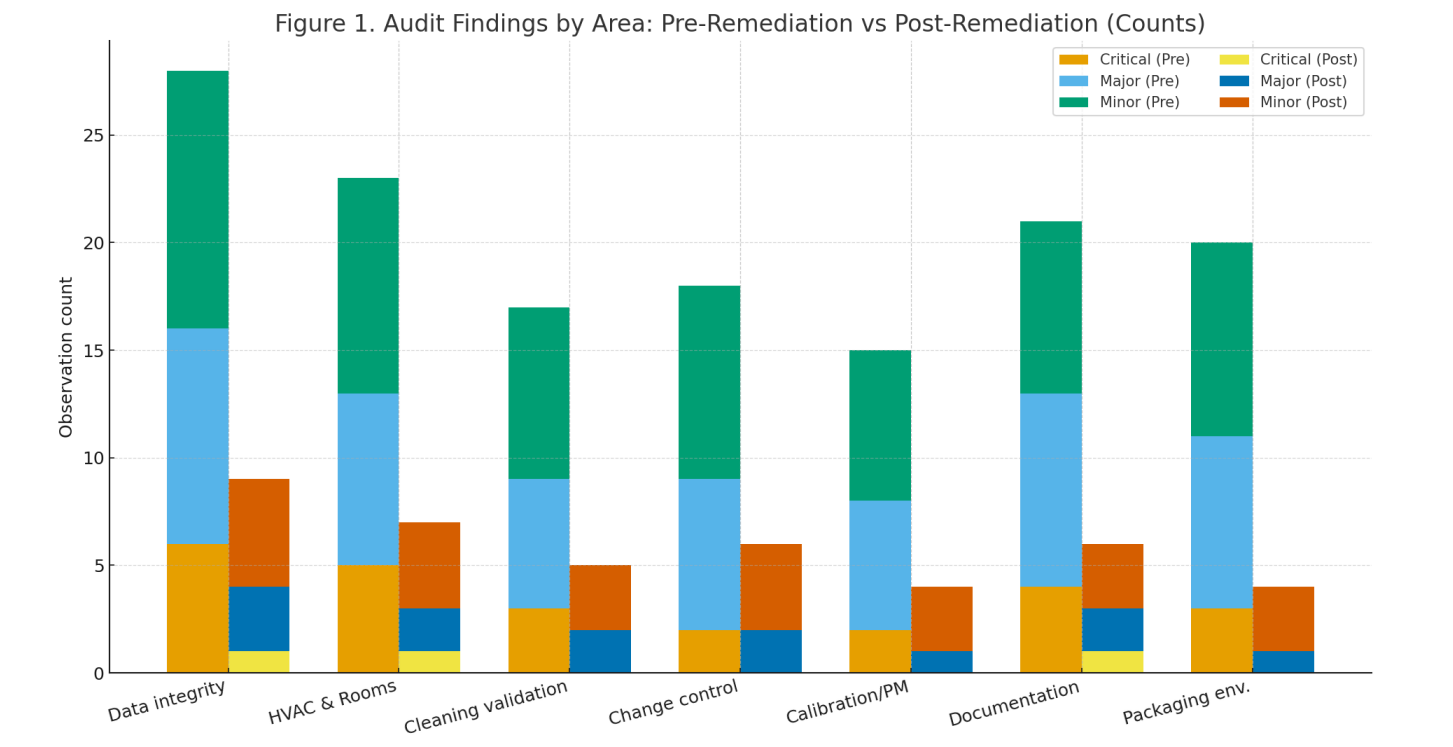
Remediation program: parallel workstreams—Quality System overhaul; HVAC rebalancing and room re-classification; equipment URS→IQ/OQ/PQ; analytical method transfer/validation; data-integrity remediation (ALCOA+); operator training with effectiveness checks; mock audit and CAPA closure.
Measurement framework:
Compliance: counts of audit observations by area and class.
Capability & release: Cpk for thickness and seal strength; time from batch completion to QP/QA disposition.
Stability: T*90 under ICH conditions; packaging leak rate.
Economics: remediation cost buckets vs benefits (scrap reduction, faster release, new CMO contracts).
Visualization: stacked bars of findings (Fig. 1), Gantt of remediation (Fig. 2), and cost–benefit waterfall (Fig. 3).
Measures
Compliance metrics: Critical/Major/Minor per audit area; % CAPA closed on time; repeat finding rate.
Quality metrics: thickness Cpk ≥1.33; residual solvent within ICH Q3C; moisture variability RSD ≤5%; seal-strength Cpk ≥1.33.
Operational metrics: release lead time (days), OTIF, deviation density (per 1,000 lots).
Economic metrics: remediation spend (HVAC/rooms, utilities, CSV, IQ/OQ/PQ, training, downtime), and annualised benefits.
Results
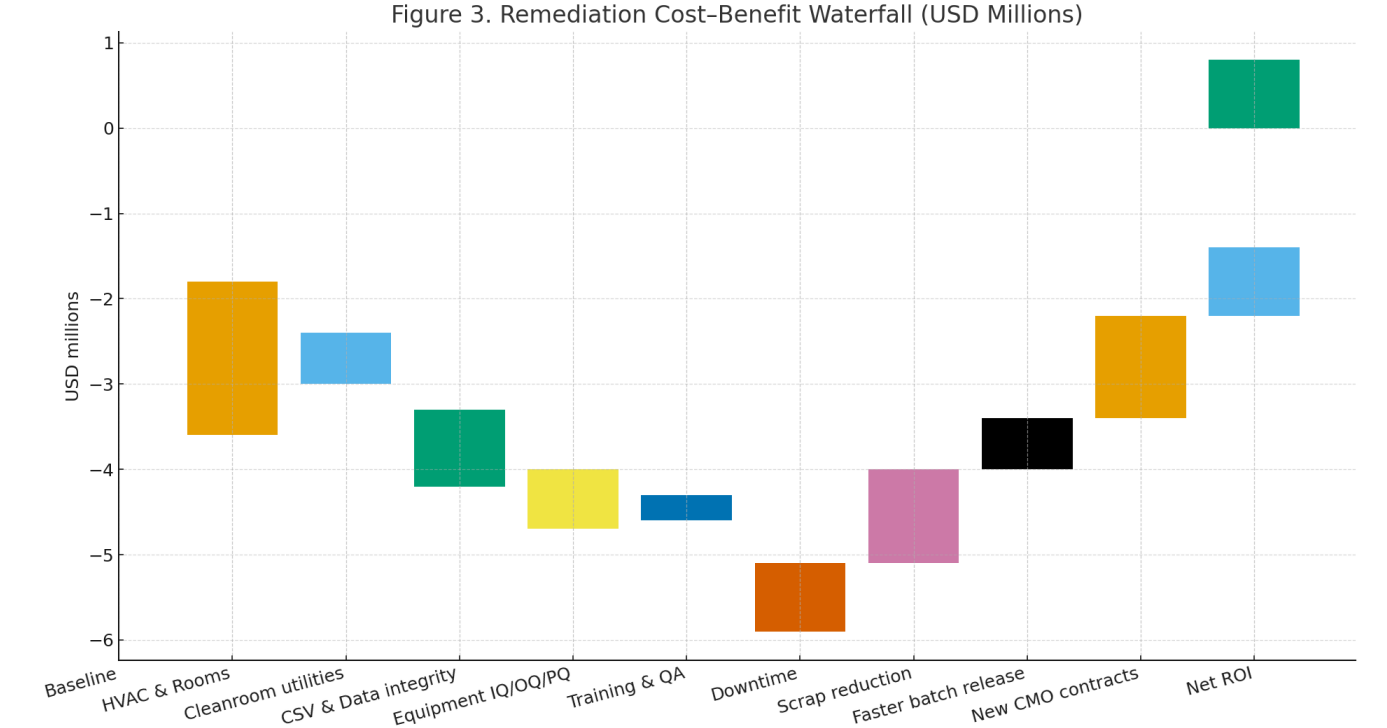
Audit outcomes. Figure 1 shows pre-remediation clusters in data integrity, HVAC/rooms, and documentation. Post-remediation, critical findings fell from 19 to 2 total, major from 54 to 13, and minor from 63 to 23 (illustrative).
Quality & release. Thickness and seal-strength Cpk rose to ≥1.33; residual solvent out-of-spec fell by ~70%; average batch disposition time shortened by 2–4 days after method validation and e-logbook deployment.
Operations. OTIF improved by ~7–10 percentage points; repeat-finding rate in follow-up audits dropped below 5%.
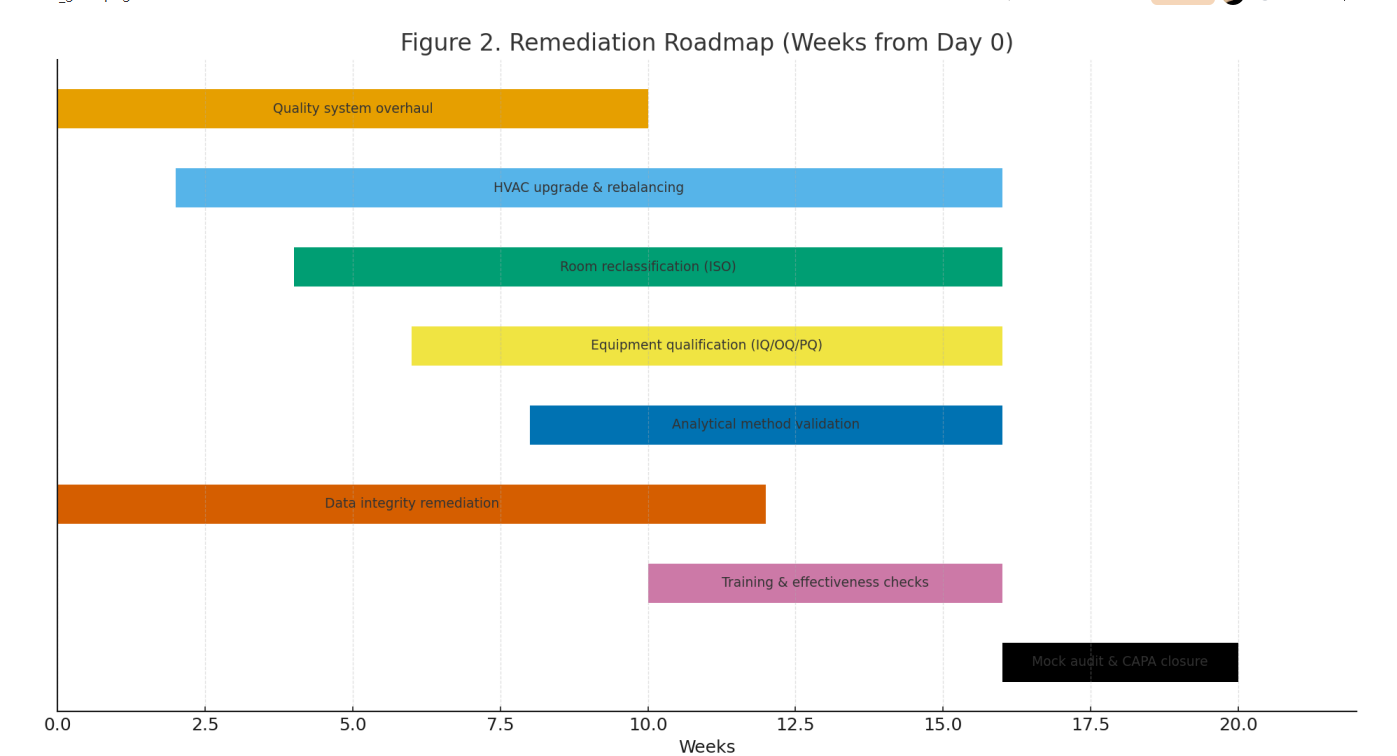
Economics. The waterfall (Fig. 3) indicates up-front costs dominated by HVAC/room works and CSV/data integrity; benefits accrue via scrap reduction, faster release, and eligibility for new CMO contracts, yielding a positive net ROI within ~18–24 months (scenario basis).
Program cadence. The roadmap (Fig. 2) completed in ~20 weeks with overlapping workstreams; mock audit closed residual CAPAs before re-inspection.
Discussion
Why non-GMP sites fail. Weak quality governance (change control/training), unqualified equipment, and HVAC/room classification mismatches violate GMP fundamentals and directly degrade ODF CQAs. What fixes it. Annex 15-aligned qualification (URS→DQ→IQ/OQ/PQ), CSV with ALCOA+ controls, and environmental rebalancing (pressure cascades, RH) stabilize the process and documentation trail [2–5].
Trade-offs. Capital and downtime are substantial; however, controlled environments and data integrity prevent chronic deviations and unlock higher-margin contracts. Sustainability. Continued Process Verification (CPV) and periodic mock audits are required to avoid regression.
Limitations. Results are scenario-based and may vary by polymer/solvent system and site starting maturity; nonetheless, the sequence generalises to most ODF facilities.
Conclusion
Non-GMP ODF facilities fail audits for predictable reasons—but with a structured, parallel remediation program that integrates quality-system rebuilds, facility upgrades, equipment qualification, and data-integrity fixes, sites can return to compliance, protect product quality, and regain commercial viability. Though expensive, remediation delivers operational stability and positive ROI when paired with disciplined CPV and governance.
References
[1] EU-GMP Vol. 4 & Annex 15: Qualification/Validation and tech transfer expectations.
[2] PIC/S PE 009: Guide to GMP for medicinal products.
[3] FDA Guidance on Data Integrity and CGMP compliance.
[4] ICH Q8/Q9/Q10: Pharmaceutical development, risk management, and quality systems.
[5] ASTM F1249/F1927: WVTR/OTR barrier testing—packaging integrity for ODFs.
[6] USP/Ph. Eur. general chapters: uniformity, residual solvents, packaging integrity.
[7] ISPE/GAMP 5: Good automated manufacturing practice and CSV.