Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
GMP-compliant oral dissolving film (ODF) manufacturing demands a validation strategy that traces user requirements (URS) through Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ)—and then into ongoing control. This paper provides a practical checklist linking CPPs (e.g., slot-die gap, flow, multi-zone drying ΔT/face velocity, exit moisture, conditioning RH/time, seal T/P/dwell) to CQAs (thickness CV%, assay RSD%, residual moisture, seal/opening force, pouch reject ppm) and shows how to evidence conformity across IQ/OQ/PQ. Three figures illustrate the V-model traceability, an IQ–OQ–PQ coverage matrix, and PQ capability indices for release-critical CQAs. [1–9]
Introduction
ODF equipment—slot-die coaters, multi-zone dryers, slitters, and form-fill-seal pouching lines—combine coating science, web handling, and packaging. Validation must prove fitness for intended use, show that operations are controlled within a proven process window, and confirm that the system can reproducibly meet product specifications. IQ confirms installation and utilities/identification; OQ challenges functions and alarms across ranges; PQ shows process capability on real product under routine controls. [2–6]
Methods
URS and risk assessment. Derive URS from quality targets and markets; perform FMEA to rank failure modes affecting dose uniformity, moisture/curl, and seal integrity. [1,6]
IQ (documents + tests). Verify equipment ID, utilities, wiring/air/vacuum, calibration state, software version & access control, and data pathways (PAT sensors → historian). Record GR&R baselines for critical gauges. [4–6,8]
OQ (ranges & challenges). Challenge gap/flow/speed, dryer ΔT & face velocity, web tension, EBR settings, vision/PAT alarms, and seal window (T/P/dwell). Include power loss, e-stop, interlocks and data-integrity tests (ALCOA+). [3–6,8–9]
PQ (process capability). Run ≥3 conformance lots at target settings; demonstrate Cp/Cpk ≥ 1.33 for release-critical CQAs and acceptable pouch reject ppm with opening-force windows. Lock recipes, limits, and reactions (SPC/EWMA with Western-Electric rules). [4–7]
Ongoing control. Periodic calibration/verification, change control, deviation/CAPA tracking, and annual review; maintain state of control with PAT dashboards and audit trails. [5–9]
Measures
Uniformity & integrity: thickness CV%, cross-web P–V (µm), assay RSD%, residual moisture (%), curl (mm).
Operations: alarm response time, interlock effectiveness, tension stability (N), PAT–lab bias and GR&R.
Packaging: seal strength (N/15 mm), opening force (N), visual defects/ppm, laminate OTR/WVTR fit.
Capability: Cp/Cpk for release-critical CQAs; OEE, scrap (%), good-meters/min. [3–9]
Results
Traceability and lifecycle control
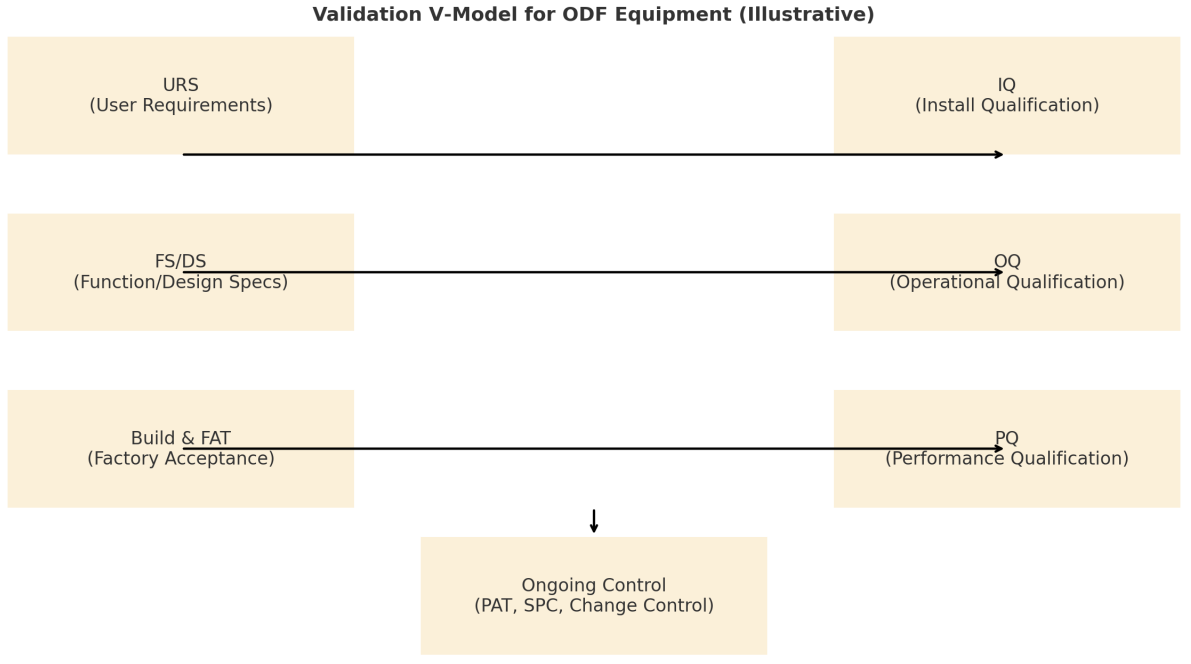
Figure 1 shows a validation V-model: URS map to IQ/OQ/PQ with ongoing control (PAT/SPC/change control) closing the loop. This ensures every URS element (e.g., moisture PAT, data integrity) has a defined verification route. [1,6–8]
Coverage across IQ–OQ–PQ
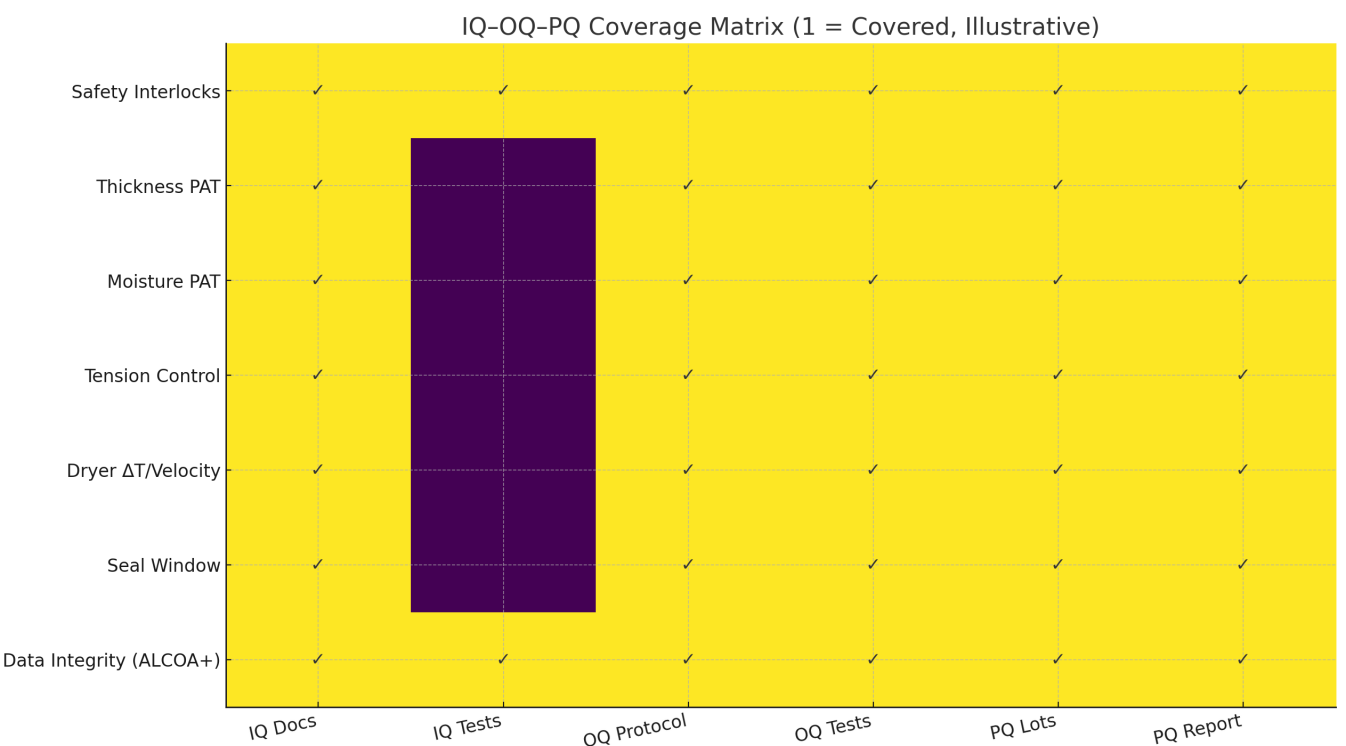
Figure 2 (matrix) indicates each URS topic has coverage: safety/interlocks and data integrity anchor all phases; process capability drivers (thickness/moisture PAT, tension, dryer design, seal window) are proven in OQ and demonstrated in PQ lots. [3–6,8–9]
PQ capability outcomes
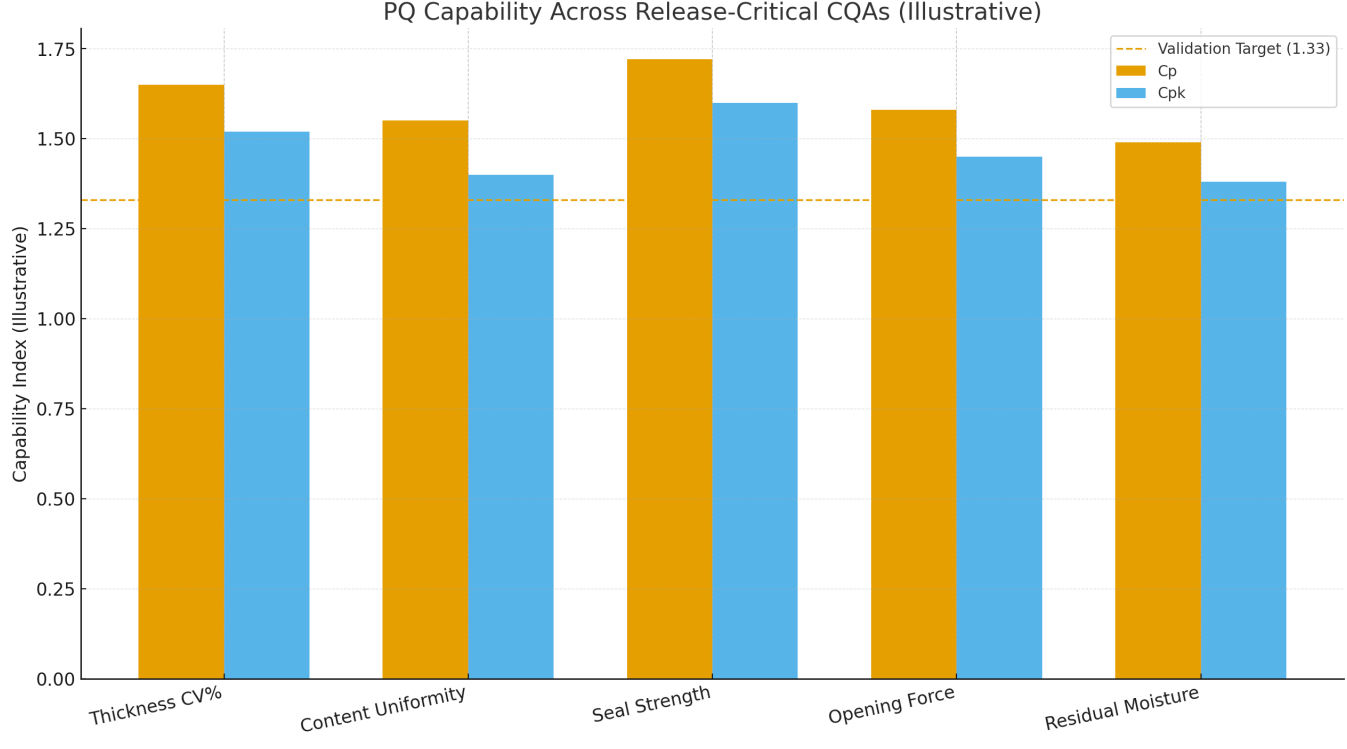
Figure 3 shows illustrative Cp/Cpk results for thickness CV%, content uniformity, seal strength, opening force, and residual moisture; all meet or exceed 1.33 with headroom, supportive of release by exception policies. [4–7]
Discussion
Checklist that passes audits
IQ essentials: asset IDs/serials, utilities drawings, calibration certificates, software validation scope, access control & audit trails, backup/restore, and as-built P&IDs.
OQ stress & alarms: verify interlocks, simulate sensor faults, push dryer zones to edges, map seal window across T/P/dwell, and confirm PAT alarms propagate to the historian with time stamps (ALCOA+).
PQ realism: product-grade materials, normal staffing, typical shift variation; demonstrate steady-state and changeover reproducibility.
CPP→CQA guardrails
Solids% vs wet gap to reach dose without inflating leveling time and curl.
Balanced drying (ΔT & face velocity) to avoid skin-over that traps solvent → moisture non-uniformity.
Edge-bead control (EBR + lip shims) to flatten cross-web thickness and reduce edge trim and assay RSD.
Seal window & laminate selection to stabilize opening force and reduce pouch rejects. [2–6,8–9]
Data integrity (ALCOA+)
Ensure attributable, legible, contemporaneous, original, accurate records; design PAT/historian so that inline thickness/moisture/vision data cannot be overwritten; periodic reviews detect drift and unauthorized changes. [8–9]
Conclusion
A robust validation program for ODF equipment starts with URS and ends with ongoing control. By proving installation, challenging operations across ranges, and demonstrating performance capability on real product, manufacturers can sustain audit-ready compliance while enabling higher speeds, lower scrap, and predictable quality.
References
Validation planning, URS traceability, and risk-based approaches for manufacturing systems.
Slot-die coating & web handling fundamentals relevant to ODF processes.
Multi-zone drying design: ΔT, face velocity, residence time, and moisture uniformity.
QbD/PAT: inline thickness/moisture/vision, GR&R, historian, and SPC/EWMA use.
CPP→CQA mapping for thin films: solids%, coat weight, tension, curl, blocking.
Packaging validation: seal window (T/P/dwell), laminate OTR/WVTR, opening-force windows.
Process capability (Cp/Cpk ≥ 1.33) and performance verification using conformance lots.
Computerized systems & data integrity: ALCOA+ expectations and audit trails.
Change control, calibration, and periodic review programs for maintaining state of control.